Competency Area 7: Using Pesticides in an Environmentally Sound Manner
PO 41. Recognize how movement of a pesticide in soil or into water may be affected by:
- Soil texture
- Erosion
- Pesticide degradation
- Pesticide persistence
- Degradation processes
- Leaching
- Precipitation runoff
- Pesticide solubility
- Pesticide adsorption
- Source of entry into the environment
A. Soil Texture
Soil type is a major factor determining how much water percolates through the soil profile and how much runs off the surface. The soil's permeability determines the potential for water transport through the soil profile. This can typically be inferred from soil texture.
Coarse-textured sands and gravels have high infiltration capacities, and water tends to percolate through the soil rather than to run off over the soil surface. Therefore, coarse-textured soils generally have high potentials for leaching of pesticides to groundwater but low potentials for surface loss to streams and lakes. Fine-textured soils such as clays and clay loams generally have low infiltration capacities, so surface runoff is relatively high compared to percolation. The potential for pesticide surface loss therefore is high and the potential for leaching is low. However, such soils may exhibit large pores formed as shrinkage cracks (when dry), worm holes or root channels. These may extend into the soil for several feet and can act as conduits for of chemicals.
B. Erosion
If pesticides are applied to the soil surface without incorporation, they are susceptible to loss through runoff and erosion during high-intensity rainfall events. Surface losses will likely result in contamination of streams, lakes, and estuaries. The potential for surface loss depends on pesticide properties, soil type, and the length of time after application. Pesticides that attach easily to soil particles or are very insoluble tend to remain close to the soil surface. At times when soils are not protected by crop canopies, high intensity rainfall may cause erosion and removal of sediment particles to streams. This is of particular concern with persistent pesticides on highly erodible soils. The highest risk period is immediately after application, before the pesticides have moved into the soil or have degraded.
C. Pesticide degradation
The rate of degradation for a pesticide typically is expressed as half-life, the period of time required for half of the original quantity to be degraded. The half-life for each compound depends on many factors, including:
- Chemical structure. Some types of chemical compounds are more easily degraded through chemical or microbial reactions than others.
- The amount of sunlight, if photochemical reactions play a role.
- Soil type. Soil properties affect pesticide degradation in many ways. In general, the higher the organic matter content and moisture-holding capacity of the soil, the higher the rate of pesticide degradation in that soil.
- Temperature. The rates of microbial and chemical reactions increase with temperature, so pesticide degradation occurs faster as the soil and air become warmer.
- Soil water content. Microbial and chemical reactions are favored by moist soil conditions, so degradation occurs fastest when soils are not too dry. When soils are water-saturated, however, oxygen supplies gradually decrease and an anaerobic environment develops. This typically slows pesticide degradation.
- Position in the soil. The upper layers of the soil profile are chemically and biologically most reactive. Once a pesticide has moved below the rooting depth, the degradation rate becomes very low because of decreased microbial activity.
D. Pesticide persistence
A low degradation rate (long half-life) indicates that the pesticide tends to persist in the environment and therefore to remain available for potential contamination of surface or groundwater.
E. Degradation processes
Most pesticides are organic compounds which degrade under typical environmental conditions. Pesticide degradation includes several types of processes:
- Microbial degradation. Soils and plants hold populations of microorganisms which derive energy from the degradation of organic compounds such as pesticides. Two important processes are distinguished: (1) mineralization, in which the compound is completely degraded to carbon dioxide (CO2 ), and (2) cometabolization, in which the chemical is transformed into other chemical compounds.
- Photochemical reactions or decomposition through exposure to sunlight. These reactions are called photolysis.
- Chemical reactions. Pesticides may react with air, water, and other chemicals in soil and plants through oxidation, reduction, and hydrolysis. For example, through a series of reactions, aldicarb degrades into several other compounds.
Degradation reactions typically produce harmless products such as carbon dioxide, but can also yield compounds which themselves are toxic, such as the aldicarb sulfoxide and aldicarb sulfone produced as intermediary products of aldicarb degradation
F. Leaching
Once groundwater becomes contaminated at concentrations which are higher than the drinking water standard, the wells tapping into this water have to be closed. Reclamation of contaminated groundwater is typically prohibitively expensive.
Groundwater originates as recharge, the water that percolates downward through soil to the depth at which all soil pores are saturated. Recharge areas may be very small or may extend over many square miles. Depending on local geology and groundwater flow characteristics, water in any given well may be recharged from the land directly adjacent to the well or from areas miles away. Shallow wells typically are recharged by water originating from adjacent land.
Recharge takes place intermittently during and immediately following periods of rainfall or snowmelt. Under wet soil conditions, recharge carries with it any pesticides that are dissolved in the soil solution. Pesticides applied in the spring may move downward rapidly, especially through coarse-textured (sandy, gravelly) soils. Once the chemical has moved beyond the root zone, it cannot be absorbed by plants and its rate of degradation is reduced, so its potential for groundwater contamination is greatly increased.
During the summer little leaching of pesticides occurs. Precipitation replenishes dry soil and is subsequently taken up by plants. Timely rains are then beneficial, because they promote vigorous plant growth and decrease susceptibility to pests. Pesticides applied in early summer have on the average a lower potential for leaching than those applied earlier in the growing season, because drier soil conditions and deeper roots typically prevent water from percolating to depths below the root zone.
G. Precipitation runoff
Water flow is the most important transport mechanism for pesticides. The assessment of the potential for pesticide loss to surface or groundwater should therefore include an evaluation of the site-specific water balance. Water is added to the soil through precipitation or irrigation, which either infiltrates into the soil or runs off the soil surface. The fraction of water that infiltrates compared to the fraction that runs off depends largely on the intensity of precipitation and the infiltration capacity of the soil. For example, if rainfall rates are high and the soil is a compacted clay loam, little water will enter the soil and most will be lost through runoff. This is especially the case when the soil is near saturation and therefore has a low capacity for absorbing additional water from precipitation. When runoff carries dissolved pesticides or those adsorbed to eroding soil particles, contamination of surface water resources can result.
Snowmelt, rain, and low evapotranspiration rates in the spring generate wet soil conditions and downward movement of water to groundwater. The potential for runoff is high during this period because the near-saturated or partially frozen soils have low water infiltration capacities. In addition, runoff and erosion often are aggravated by the lack of crop canopy which protects the soil from direct raindrop impact. During the summer, high rates of evaporation and plant water uptake may reduce soil water storage, leaving none to percolate downward. Summer rains only partially recharge the soil profile, and the soil's moisture holding capacity is typically not exceeded. Except for during high-intensity thunderstorms, runoff and erosion potentials are generally low during the summer because of the protective crop canopy and the higher water absorption capacity of the soil. In the late fall, evapotranspiration rates strongly decrease, and groundwater recharge occurs when the moisture-holding capacity of the soil is exceeded. Runoff and erosion potentials also increase during this period. Because late fall and especially early spring tend to be the seasons of greatest runoff, erosion, and groundwater recharge, pesticide management should aim at keeping soil pesticide concentrations low at these times of the year.
H. Pesticide solubility
The tendency of a chemical to dissolve in water is expressed by its solubility. Pesticides with solubilities below the threshold value of 30 mg/l are considered to have relatively low potentials for leaching. Pesticides with solubility values higher than 30 mg/l may have a high leaching potential if the degradation rate and the soil adsorption coefficient are low. If poorly soluble pesticides are applied to soil but not incorporated, they have a high potential for loss through runoff or erosion.
I. Pesticide adsorption
A pesticide's tendency to be adsorbed by soil is expressed by its adsorption coefficient:
![]()
High K(oc) values indicate a tendency for the chemical to be adsorbed by soil particles rather than remain in the soil solution. Since pesticides bond mainly to soil organic carbon, the division by the percentage organic carbon in soil makes the adsorption coefficient a pesticide-specific property, independent of soil type. Adsorption coefficients less than 500 indicate a considerable potential for losses through leaching.
J. Source of entry into the environment
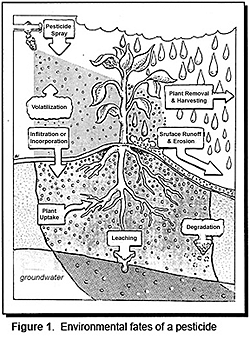
After a pesticide is applied to a field, it may meet a variety of fates (Figure 1). Some may be lost to the atmosphere through volatilization, carried away to surface waters by runoff and erosion, or broken down in the sunlight by photolysis. Pesticides which have entered into soil may be taken up by plants (and subsequently removed), degraded into other chemical forms, or leached downward, possibly to groundwater. The remainder is retained in the soil and continues to be available for plant uptake, degradation, or leaching. If a pesticide is not readily degraded and moves freely with water percolating downward through the soil, it may reach groundwater. If, however, the pesticide is either insoluble or tightly bound to soil particles, then it is more likely to be retained in the upper soil layers and small amounts may be lost to surface waters through runoff or erosion. Pesticide fate is also affected by a multitude of weather, soil, and geologic conditions, in addition to characteristics of the pesticide itself. In order to effectively manage pesticides, the properties of the chemicals, the environment, and their interaction need to be studied |
*This information was adapted from “Pesticide Management for Water Quality; Principles and Practices” Van Es, HM and Trautmann, NM. 1990. See http://psep.cce.cornell.edu/facts-slides-self/facts/pestmgt-water-qual-90.aspx for more detailed information.
Quick Links
- Competency Area 1: Integrated Pest Management (IPM)
- Competency Area 2: Weed Management
- Competency Area 3: Management of Infectious Plant Diseases
- Competency Area 4: Management of Arthropods
- Competency Area 5: Pesticide Formulations and Labels
- Competency Area 6: Management of Pesticide Resistance
- Competency Area 7: Using Pesticides in an Enviromentally Sound Manner
- Competency Area 8: Protecting Humans from Pesticide Exposure