Competency Area 4: Nutrient Sources, Analyses, Application Methods
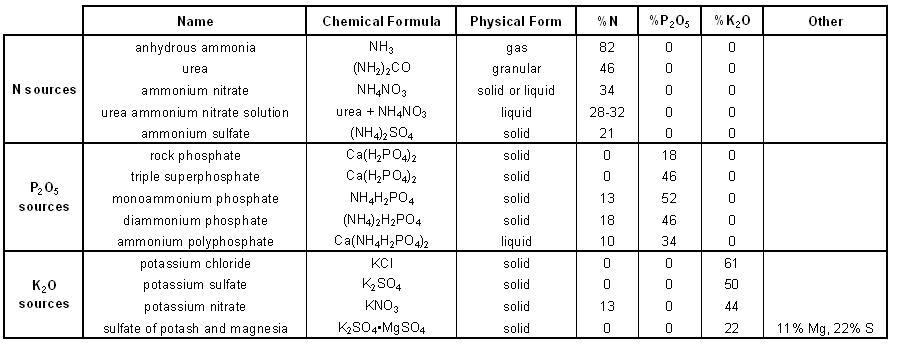
PO 28. Describe the physical form and analysis of each of the following potassium sources.
- Potassium chloride
- Potassium sulfate
- Potassium nitrate
- Potassium magnesium sulfate
- Potassium chloride: KCl
- Fertilizer grade: Contains 60-63% K2O
- Physical form: solid
- Most abundantly used potassium fertilizer
- Often referred to as Muriate of Potash
- Water soluble source of K
- Potassium sulfate: K2SO4
- Fertilizer grade: 50% K2O
- Physical form: solid
- Potassium nitrate: KNO3
- Fertilizer grade: contains about 13% N, 44% K2O
- Physical form: solid
- Potassium magnesium sulfate: K2SO4•MgSO4
- Contains about 22% K2O, 11% Mg, 22% S
- Physical form: solid, water-soluble
- Good source of Mg and S
- Often referred to as Sul-Po-Mag or K-Mag
Fertilizer Overview